 
They are 3d- series (1st transition series), 4d –series (2 nd transition series), 5d – Series (3 rd transition series) & 6d – series (4 th transition series).Įlements which kept kept separately under the table in two seperate rows in the Modern periodic table are called lanthanides and actindes. Transition elements are classified into four series of elements. General electronic configuration of transition elements is ns 2 (n-1)d 1-10. In transition elements the valency electrons goes to d – sub shell. Elements present between the group -2 and group-13 in the modern periodic table are the transition elements. This group elements are also known as inert gas elements.Įlements which contains incomplete penultimate ((n-1) : d -sub shell) shell with electrons are called transition elements. These elements have eight electrons (octet configuration) in their valence shell. Their general electronic configuration is ns 2np 6. Helium, Neon, Argon, Krypton, Radon and Xenon are elements of this. This group elements are also called as halogens. 
These elements have seven electrons in their valence shell. Their general electronic configuration is ns 2np 5. This group elements are also known as chalcogens.įluorine, Chlorine, Bromine, Iodine and Astatine are the elements of this group. These elements have six electrons in their valence shell. Their general electronic configuration is ns 2np 4. Oxygen, Sulphur, Selenium, Tellurium and Polonium are the elements of this group. These group elements are also called as pnictogens. These elements have five electrons in their valence shell. Their general electronic configuration is ns 2np 3. Nitrogen, Phosphorous, Arsenic, Antimony and Bismuth are the elements of this group. These elements have four electrons in their valence shell. Their general electronic configuration is ns 2np 2. These group elements mostly form covalent compounds.Ĭarbon, Silicon, Germanium and Tin are the elements of this group. These elements have three electrons in their valence shell. 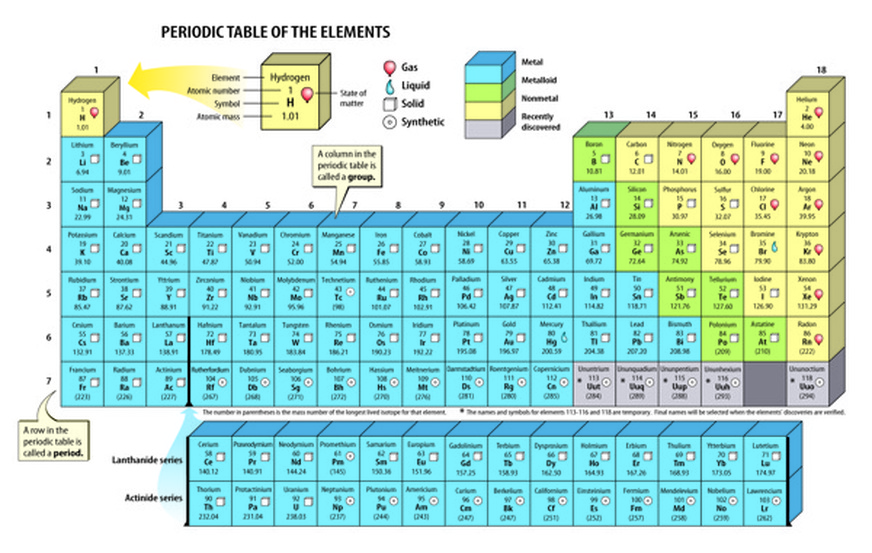
Their general electronic configuration is ns 2np 1. These elements obtained from earth crust because of these reasons these elements are called as alkaline earth metals.īoron, Aluminium, Gallium, Indium and Thallium are the elements of this group. The hydroxides of these group elements are basic in nature and soluble in water. These elements have two electrons in their valence shell. Their general electronic configuration is ns 2. This group elements are also known as alkali metals as the hydroxides of these group elements are basic in nature and soluble in water.īeryllium, Magnesium, Calcium, Strontium, Barium and Radium are the elements of this group. These elements have one electron in their valence shell. Their general electronic configuration is ns 1. Lithium, Sodium, Potassium, Rubidium, Cesium and Francium are the elements of this group. The elements of group -1, group-2 and group-13 to group-17 are the representative elements. The elements in the modern periodic table were classified into four types. Elements in the modern periodic table are arranged in 7 periods and 18 groups.įilled orbitals and number of elements in different periods: PeriodĮlements were classified into groups based on the number of valence electrons.Īccording to IUPAC nomenclature the 18 groups in modern periodic table are numbered as 1 to 18.Horizontal rows in the periodic table are called periods and vertical columns in the table are called groups.Elements are arranged in the increasing order of atomic numbers.The main features of modern periodic table: Atomic number is the basis for modern periodic table.Ītomic number is the number of protons in the nucleus, it is also equal to the number of electrons in the atom. The Modern Periodic Law is stated as: “Properties of elements are a periodic functions of their atomic number”. So Mendeleev’s Periodic law was modified and atomic number was adopted as the basis of the Modern Periodic Table. In 1913, Henry Moseley showed that the atomic number of an element is a more fundamental property than its atomic mass. The systematic arrangement of elements into groups and periods is called periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
